Thursday, December 6, 2012
Joy and wonder - the science behind mayonnaise and other things
I have been reading a curious book on soft matter by Michel Mitov that came out earlier this year called Sensitive Matter: foams, gels, liquid crystals, and other miracles. Dr. Mitov is Director of Research at the National Center for Scientific Research (CNRS) in France. It is written for non-scientists, and thus uses words and graphics to elegantly make the complex simple.
His seven-page first chapter discusses mayonnaise. It describes how the amphiphilic lecithin molecules in egg yolk act as mediators to create a stable emulsion from oil and water based ingredients. Those molecules have a head which likes water (but not oil) and two tails which like oil (but not water).
He illustrates a lecithin molecule with a simple schematic, rather than a more detailed chemical formula (which only would be understandable to chemists).
In the sixth chapter he describes how other amphiphilic molecules are responsible for making a stable foam - the bubbles in champagne. Lecithin also shows up in the second chapter on digestion, where bile salts make cholesterol into an emulsion. On page 14 he shows us a ternary phase diagram for bile salts, lecithin, and cholesterol.
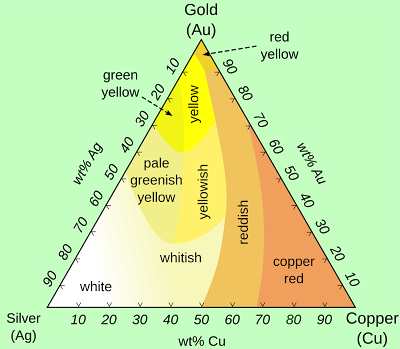
I was surprised to see that diagram, since I’m much more familiar with seeing ternary diagrams for three elements, like the one shown above that describes the colors for alloys of very expensive gold, less expensive silver, and inexpensive copper used to make jewelry.
The mayonnaise jar and ternary diagram images both came from Wikimedia Commons.
Subscribe to:
Post Comments (Atom)


No comments:
Post a Comment